
In many streams and lakes, "phosphorus is the nutrient that is the most scarce, and therefore it is often the most important nutrient," said Dan Obenour, a professor of civil, construction and environmental engineering at North Carolina State University. Such blooms, which occur in other drinking-water lakes across the country as well, are often caused or exacerbated by phosphorus from fertilizers and livestock waste flowing into waterways. In August 2014, the city of Toledo, Ohio, had to warn citizens against using the city's water due to a toxic algae bloom. Human phosphorus use has created problems for wildlife and people, alike. According to a 2009 study in the Clinical Journal of the American Society of Nephrology, higher blood-phosphorus levels indicate higher rates of calcification of the coronary arteries. Phosphorus can be used as a warning signal for heart disease.By 3.5 billion years ago, the element was abundant on the planet, the study found. Meteorites may have brought phosphorus to Earth, according to a 2013 study in the journal Proceedings of the National Academy of Sciences.Strike-anywhere matches can ignite on any surface because they contain a small amount of white phosphorus built in to the match head.Increasingly rare and expensive phosphorus would throw the global agriculture system into disarray, experts worry. Mineral reserves of phosphorus are estimated to last between a few decades and 300 years at the most. 
Earth may be approaching " peak phosphorus," after which the element will be harder and harder to mine.There are about 26.5 ounces (750 grams) of phosphate in the average human body, mostly in the bones, according to the RSC.It's also part of adenosine triphosphate, or ATP, the molecule that stores and releases energy to allow cells to function. As phosphate, a charged molecule, it combines with sugar to form the backbone of DNA. Despite its fiery properties, phosphorus is crucial to life. The most common use, however, is in fertilizers, according to the RSC. The non-illicit uses of phosphorus include steel-making and the production of flares. It explodes when combined with other compounds such as chlorine, sodium and ammonium nitrate, according to California's Office of Environmental Health Hazard Assessment, which flags red phosphorus as one of the dangerous ingredients used in making methamphetamines. When exposed to enough heat (500 F or 260 C), it will ignite. In certain combinations, though, red phosphorus is still very dangerous. Red phosphorus is made by heating white phosphorus under controlled conditions. The friction of the match against the red phosphorous transforms a little bit of the red phosphorus into white phosphorus, providing the ignition needed to light the match, according to Michigan State University's Science Theater. Red phosphorus is far more stable at room temperature - in fact, it's found on the side of any box of safety matches. 
Inhalation or contact with the skin is toxic, causing burns that can quickly turn fatal. According to the Centers for Disease Control and Prevention (CDC), it smells like matches or garlic. As a result, white phosphorus is used in fireworks and weaponry. It's also capable of self-igniting in air once the temperature reaches about 86 F (30 C) the only safe storage is under water. White phosphorus is waxy and gives off a slight glow in air, according to the Royal Society of Chemistry. Less common is black phosphorus, which is made of atoms arranged in a ring structure and looks a bit like the graphite at the point of a pencil. The most common forms are white phosphorus, made up of phosphorus atoms arranged like a tetrahedron (a four-sided pyramid), and red phosphorus, a solid but non-crystalline form of the element. The world phosphorus comes from a Greek word meaning "bearer of light," and this element delivers on that promise. There you can find the metals, semi-conductor(s), non-metal(s), inert noble gas(ses), Halogens, Lanthanoides, Actinoids (rare earth elements) and transition metals.(Image credit: Greg Robson/Creative Commons, Andrei Marincas Shutterstock) Element of light Please note that the elements do not show their natural relation towards each other as in the Periodic system. The unity for atomic mass is gram per mol. The lightest chemical element is Hydrogen and the heaviest is Hassium. The chemical elements ofįor chemistry students and teachers: The tabular chart on the right is arranged by Atomic mass (weight). This list contains the 118 elements of chemistry. 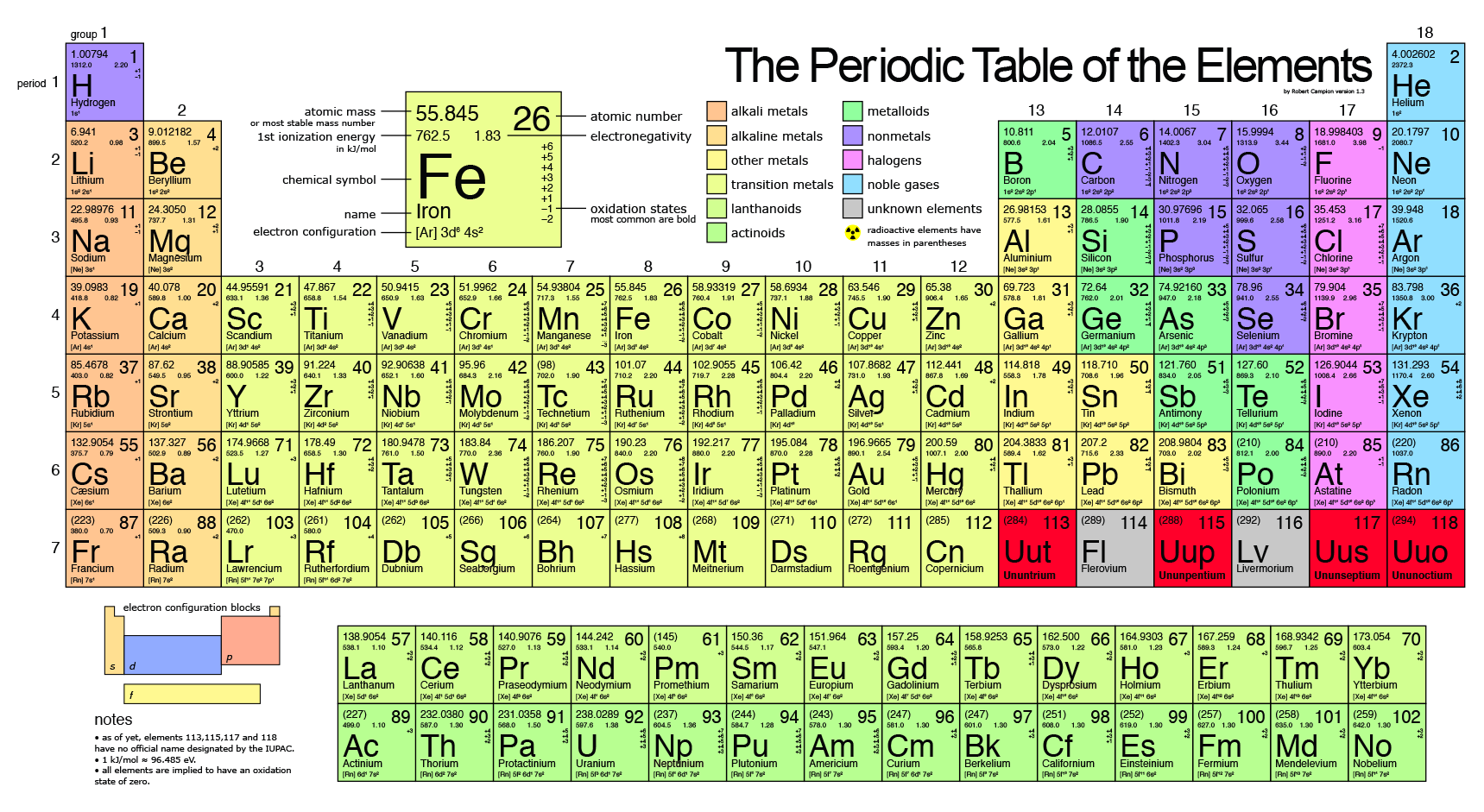
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
